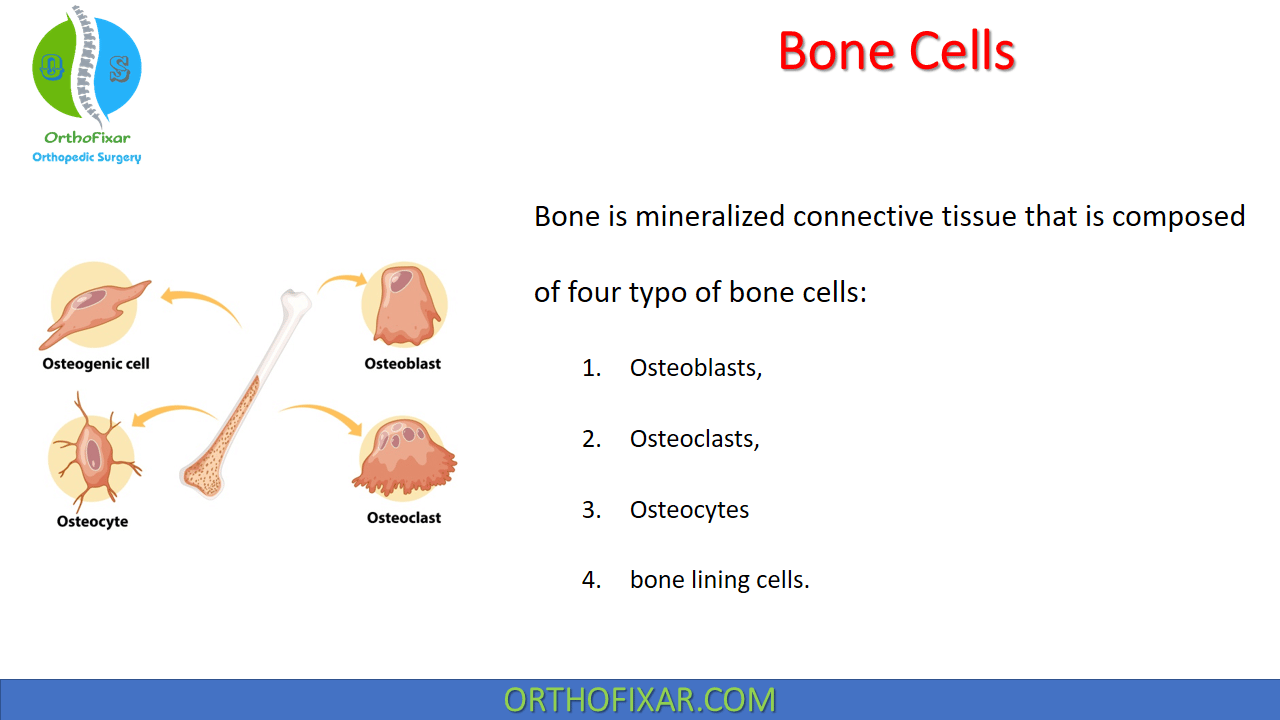
Bone is mineralized connective tissue that is composed of four typo of bone cells: Osteoblasts, Osteoclasts, Osteocytes and bone lining cells.
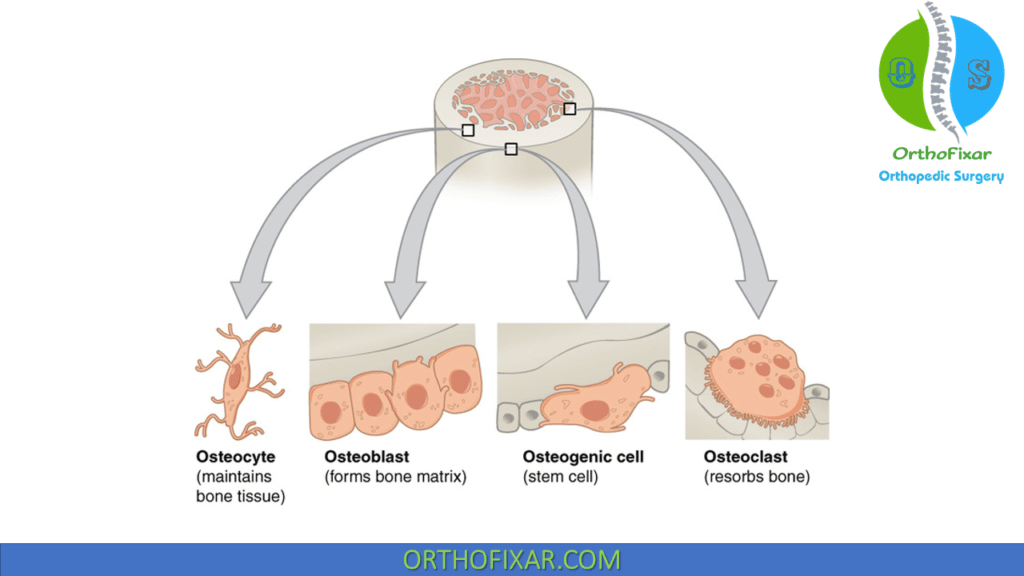
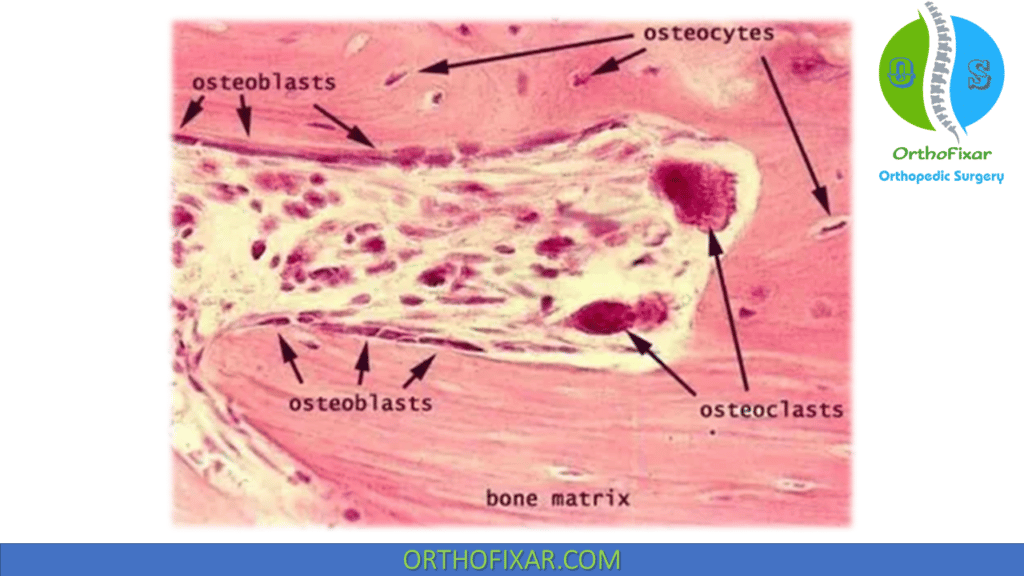
Osteoblasts
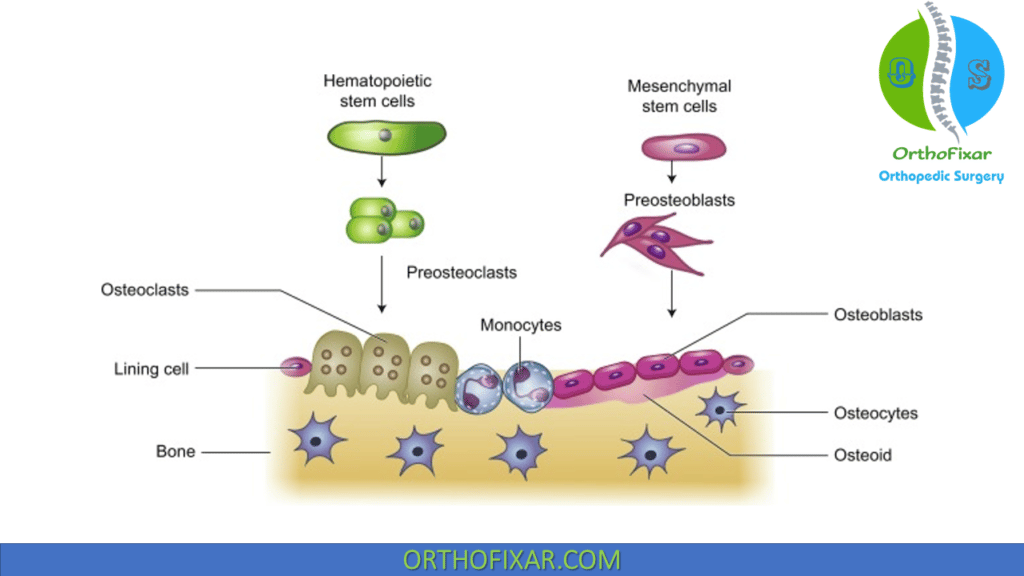
Osteoblast is the bone cells that are responsible for bone forming, they appear as cuboid cells aligned in layers along immature osteoid.
Osteoblast activity stimulated by intermittent exposure to parathyroid hormone (PTH). While its activity inhibited by tumor necrosis factor (TNF)-α.
Origin and Differentiation:
Osteoblasts bone cells are derived from undifferentiated mesenchymal stem cells. More metabolically active osteoblasts bone cells are located at the bone surface, while less active bone cells are located in more central bone.
Differentiation stimulated by:
- Bone morphogenetic protein (BMP): Stimulates Mesenchymal cells to become osteoprogenitor cells.
- Core-binding factor α-1 stimulates differentiation.
- β-Catenin stimulates differentiation into osteoblasts, with resulting intramembranous bone formation.
- Platelet-derived growth factor (PDGF).
- Insulin-like growth factor (IGF).
See Also: Bone Formation
The Osteoblasts functions are:
- Forming Bone.
- Regulating osteoclasts activity.
Osteoblasts produce the following:
- Alkaline phosphatase.
- Osteocalcin (stimulated by 1,25 dihydroxyvitamin D).
- Type I collagen.
- Bone sialoprotein.
- Receptor activator of nuclear factor (NF)-κB ligand (RANKL).
- Osteoprotegrin: binds RANKL to limit its activity.
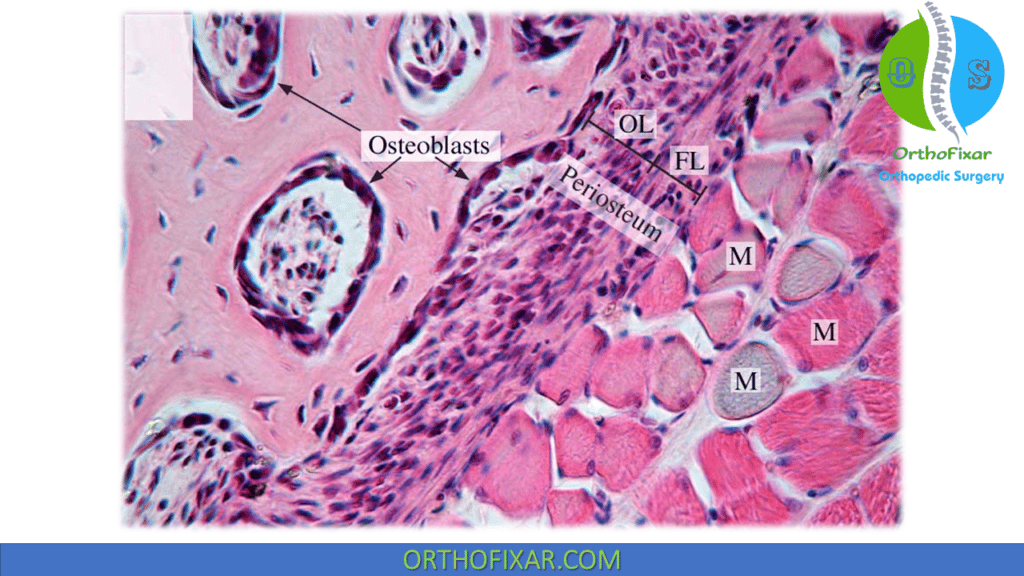
Receptor Types on Osteoblasts bone cells:
| Receptor Type | Effects |
|---|---|
| Parathyroid hormone PTH | – Releases a secondary messenger (exact mechanism unknown) to stimulate osteoclastic activity – Activates adenylyl cyclase |
| 1,25(OH)2 vitamin D3 | – Stimulates matrix and alkaline phosphatase synthesis and production of bone-specific proteins (e.g., osteocalcin) |
| Glucocorticoids | – Inhibits DNA synthesis, collagen production, and osteoblast protein synthesis |
| Prostaglandins | – Activates adenylyl cyclase and stimulates bone resorption |
| Estrogen | – Has anabolic (bone production) and anticatabolic (prevents bone resorption) properties – Increases mRNA levels for alkaline phosphatase – Inhibits activation of adenylyl cyclase |
Osteoclast
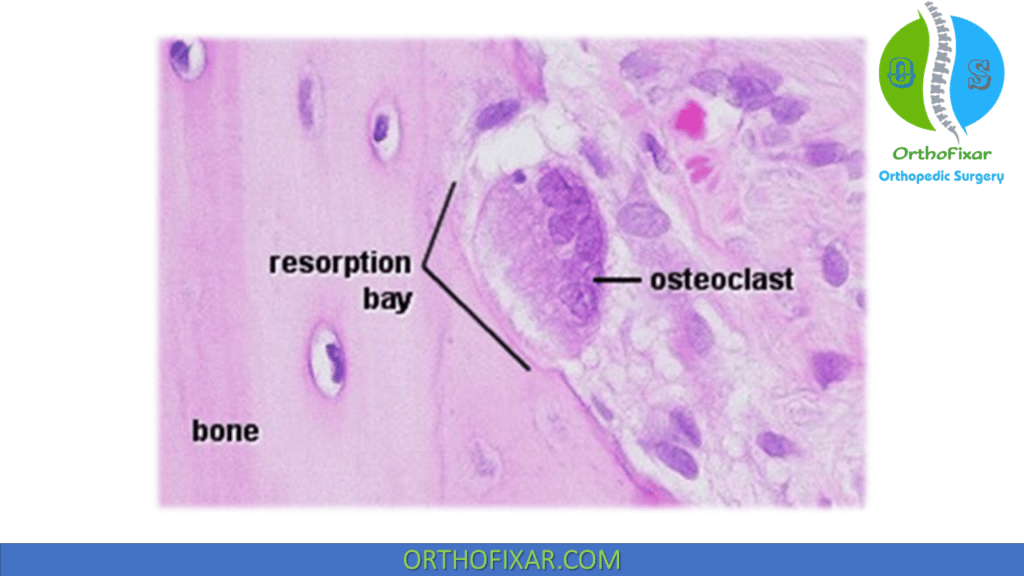
Osteoclast is the bone cells that are responsible for bone resorption. They are a multinucleated irregular giant cells.
Osteoclasts are derived from hematopoietic cells in macrophage lineage. Monocyte progenitors fuse together to form mature multinuclear giant cells.
Osteoclast functions are:
- Bone resorption:
- Bone resorption occurs in Howship lacunae.
- Osteoclasts have a ruffled (brush-like) border and surrounding clear zone.
- Border consists of plasma membrane enfoldings that increase surface area for resorption.
- Cathepsin K: it is one major proteolytic enzyme that digests organic matrix at ruffled border.
- Synthesize tartrate-resistant acid phosphate: Secreted by osteoclasts to lowers the Ph and increases the solubility of hydroxyapatite crystals
- Bind to bone surfaces through cell attachment proteins (integrins): Integrins include αVβ3, αVβ5, α2β1, αVβ1.
Osteoclasts are activated by IL-1 and RANKL, while they are inhibited by IL-10 and Calcitonin
Osteoblasts express RANKL, which acts as follows:
- Binds to receptors on osteoclasts.
- Stimulates differentiation into mature osteoclasts.
- Increases bone resorption.
IL1 is found in membranes surrounding loose total joint implants.
Bisphosphonates
Bisphosphonates inhibit osteoclastic bone resorption by preventing osteoclasts from forming ruffled border and producing acid hydrolases.
It’s is categorized into two classes on the basis of presence or absence of a nitrogen side group:
- Nitrogen-containing bisphosphonates:
- It has up to 1000-fold more potent in their antiresorptive activity.
- Zoledronic acid and alendronate.
- Inhibit protein prenylation within the mevalonate pathway, blocking farnesyl pyrophosphate synthase.
- Results in a loss of guanosin triphosphatase (GTPase) formation, which is needed for ruffled border formation and cell survival.
- Non–nitrogen-containing bisphosphonates:
- Metabolized into a nonfunctional adenosine triphosphate (ATP) analogue, inducing apoptosis.
- Decreases skeletal events in multiple myeloma.
- Associated with osteonecrosis of the jaw.
Orthopedic uses of Bisphosphonate include the following cases:
- Spine:
- Reduced rate of spinal fusion in animal model.
- Withholding bisphosphonate is recommended after surgery.
- Hip and knee:
- Safe for use in cementless hip arthroplasty and cemented knee arthroplasty;
- May decrease rate of acetabular component subsidence.
- Fracture healing:
- No good data to recommend for or against use.
See Also: Bone Metabolism
Osteocytes
Osteocytes are bone cells that were former osteoblasts surrounded by newly formed matrix. They make up 90% of the bone cells in the mature skeleton.
Osteocytes cell has high nucleus/cytoplasm ratio. It has a long interconnecting cytoplasmic processes projecting through the canaliculi, long Processes are used to communicate osteocytes with other bone cells.
Osteocytes stimulated by calcitonin, while inhibited by PTH.
Osteocytes functions include:
- Osteocytes maintain bone and cellular matrix.
- Regulation of calcium and phosphorous concentrations in bone.
- Sclerostin secreted by osteocytes helps negative feedback on osteoblasts’ bone deposition:
- Differentially regulated based on mechanical loading, with decreased sclerostin in areas of concentrated strain.
- Downregulation is associated with increased bone formation (via sclerostin antibody).
- Potential for use in fracture healing, bone loss, implant osseous integration, and genetic bone diseases via upregulating sclerostin.
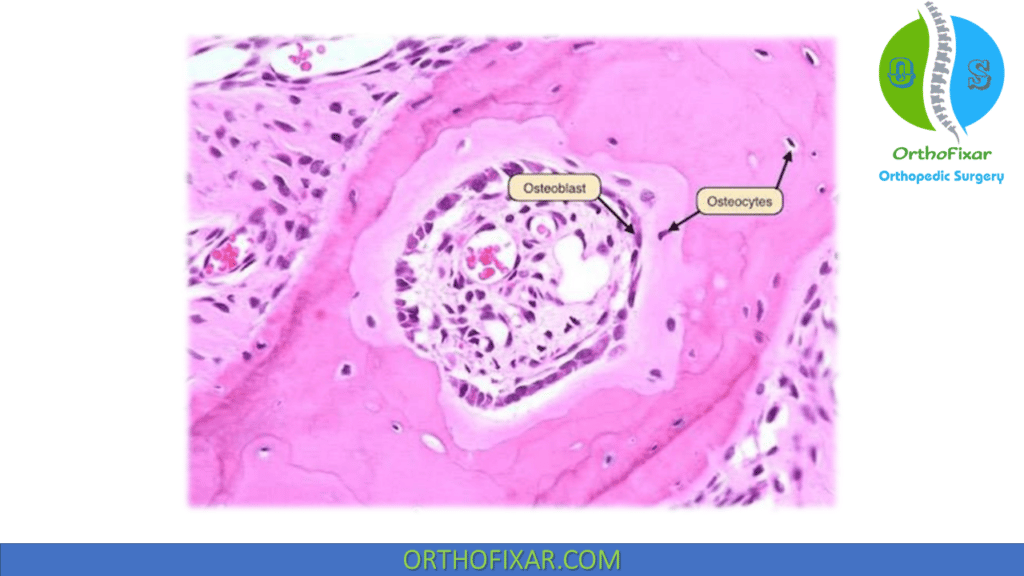
Osteoprogenitor cells
Osteoprogenitor cells originate from mesenchymal bone marrow stem cells. They differentiate into different type of bone cells based on oxygen tension:
- Osteoprogenitor cells become osteoblasts under conditions of low strain and increased oxygen tension
- Osteoprogenitor cells become cartilage under conditions of intermediate strain and low oxygen tension.
- Osteoprogenitor cells become fibrous tissue under conditions of high strain.
Bone Matrix
Bone matrix consists of organic and Inorganic components.
Organic Components:
Forms 40 % of dry weight of the bone.
- Collagen: it forms 90 % of organic components. Primarily type I that provides tensile strength of bone.
- Proteoglycan: it consists of glycosaminoglycan-protein complexes. It provides compressive strength of the bone.
- Matrix proteins (noncollagenous):
- Osteocalcin: Inhibited by PTH and stimulated by 1,25-dihydroxyvitamin D3, it can be measured in serum or urine as a marker of bone turnover.
- Osteonectin: it’s secreted by platelets and osteoblasts, and has a role in regulating calcium or organizing mineral in matrix.
- Osteopontin: It’s a cell-binding protein.
- Growth factors and cytokines: these include IL-1, IL-6, IGF, TGF-beta, BMPs.
Inorganic Components:
Inorganic (mineral) components: forms up to 60% of dry weight of bone, they include:
- Calcium hydroxyapatite [Ca10(PO4)6(OH)2].
- Calcium phosphate.
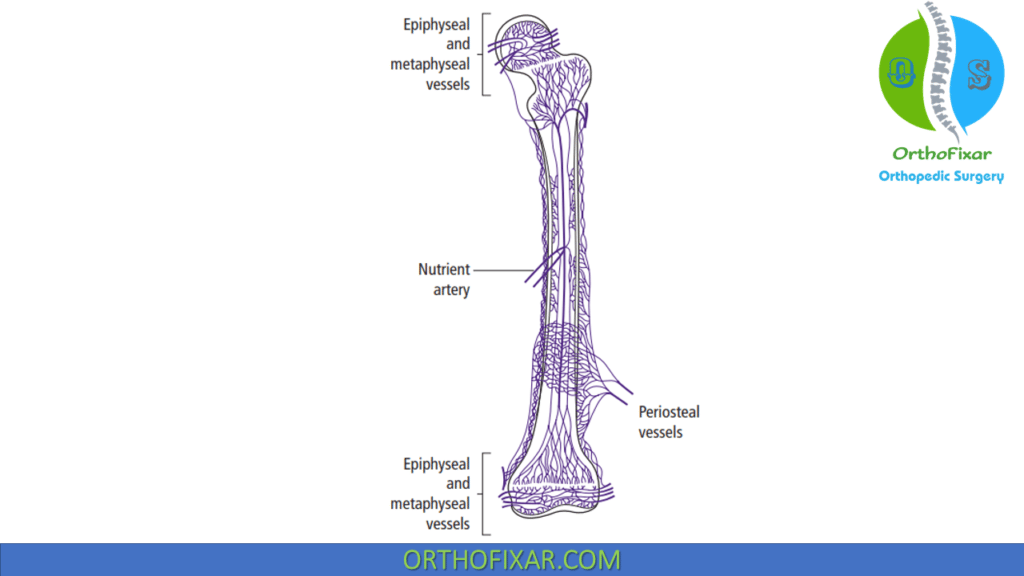
Bone Circulation
Bone receives 5% to 10% of the cardiac output. Bone Circulation main supply comes from 3 main sources:
1. Nutrient artery system:
Nutrient artery system branches from systemic arteries, enter the diaphyseal cortex through the nutrient foramen. It provides blood supply to the inner two thirds of the mature diaphyseal cortex is via the haversian system.
Blood pressure (BP) in the nutrient artery system is high.
60% of cortical bone vascularized by nutrient arteries
2. Metaphyseal-epiphyseal system
Metaphyseal-epiphyseal system Arises from the periarticular vascular plexus.
3. Periosteal system
Periosteal system consists mostly of capillaries that supply the outer third (at most) of the mature diaphyseal cortex.
Blood pressure in the periosteal system is low.
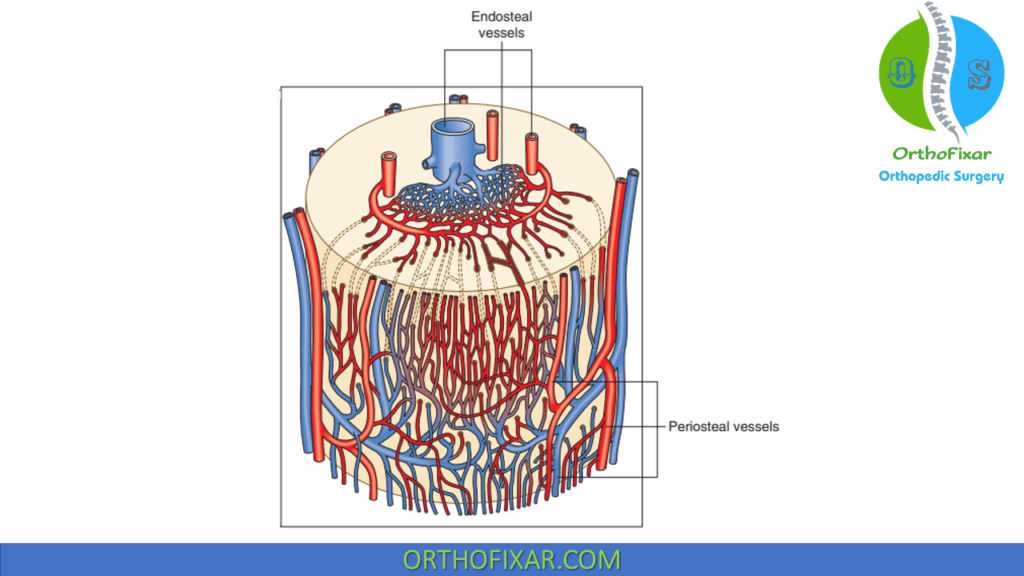
Direction of flow
Mature bone:
- Arterial flow in is centrifugal (inside to outside) due to the high pressure nutrient artery system and the low pressure periosteal system.
- When fracture disrupts the nutrient artery system, the periosteal system pressure predominates and blood flow is centripetal (outside to inside).
Immature developing bone: Flow in is centripetal because the highly vascularized periosteal system is the predominant component.
Venous flow in mature bone is centripetal.
Cortical capillaries drain to venous sinusoids, which drain to the emissary venous system.
References & More
- Millers Review of Orthopaedics -7th Edition Book.
- Basic Orthopaedic Sciences: The Stanmore Guide Book by Manoj Ramachandran.
- Campbel’s Operative Orthopaedics 12th edition Book.
- Florencio-Silva R, Sasso GR, Sasso-Cerri E, Simões MJ, Cerri PS. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. Biomed Res Int. 2015;2015:421746. doi: 10.1155/2015/421746. Epub 2015 Jul 13. PMID: 26247020; PMCID: PMC4515490.
- Mohamed AM. An overview of bone cells and their regulating factors of differentiation. Malays J Med Sci. 2008 Jan;15(1):4-12. PMID: 22589609; PMCID: PMC3341892.
Closed.